Circadian Rhythms
A call to arms to create spaces with brighter days and darker nights
Circadian rhythm is a 24-hour pattern that is found in all life, generally mediated by a central clock. Plants use their clocks to coevolve with their predators [1] and their pollinators [2]. Rodents use their clocks to determine food availability, safety and mating time [3]. All forms of life utilize their clocks for maximizing resource efficiency and survival [5].Within the human body, each cell has a clock [4]. There exists a direct link between a novel photosensitive cell in the retina and the region of the brain where the central clock is located [6], pinpointing light as the main input to reset and synchronize our clocks each day [7]. Considering life on earth evolved with the predictable solar cycle, it makes sense that light would be our primary tool to synchronize these circadian clocks.
Technological developments in recent history have broken the relationship between sun and people - we now spend more than 90% of our time indoors primarily under electric light [8]. Understanding that light is the primary synchronization tool, we now have the ability to influence our rhythms with a flip of a switch. However, these interior lighting cues are often inappropriate for the biological system: too dim to be considered daytime and too bright to be considered night [9]. Said a different way, we spend 90% of our time in a constant twilight that is neither day nor night. This constant twilight has led to circadian desynchrony, which according to a recent study affects 87% of the day-working population to some degree [10]. The downstream consequences of circadian desynchrony are quite severe, as they have been shown to lead to increased risk of obesity [11], metabolic disease, cardiovascular disease, addiction, Alzheimer’s disease and certain types of cancer [10,12].
In short, it is understood that today’s electric light is a significant part of the problem. The solution to this problem is to better delineate day versus night in the spaces where we spend our time, for example, by creating a built environment with biologically brighter days and darker nights [13].
However, the lighting measurements for visual lighting is inadequate for quantifying the circadian effectiveness of light [14]. It has been 18 years since Dr. Bud Brainard published evidence for a novel circadian photoreceptor, now known as “intrinsically photosensitive retinal ganglion cells” (ipRGCs) [15], that facilitate the syncing of the circadian rhythms. The industry is doing its best to put in place metrics to quantify circadian effectiveness. Currently there are two main metrics for quantifying this circadian effectiveness, the melanopic lux model [14] and the circadian stimulus model [16].
Circadian rhythms are kept in sync by various cues, including light which the body responds to in a way that is facilitated by intrinsically photosensitive retinal ganglion cells (ipRGCs), the non-image- forming photoreceptors of the eyes.
Circadian modelsThe Equivalent Melanopic Lux (EML) model was proposed by Lukas et al. and the Circadian Stimulus (CS) model was developed by the Lighting Research Center. The International Commission on Illumination (CIE) has also proposed a metric for based on the same biological model as EML, but scaled differently [17]. Mathematically, 1 EML = 0.91 Melanopic Daylight Equivalent D65.Both CS and EML models are based on spectral power distribution rather than simply the visual intensity. Note that both models consider the light entering the eye at the vertical plane. This is a departure from lighting for visual needs were the surface where a task is conducted or the target to be seen is considered.
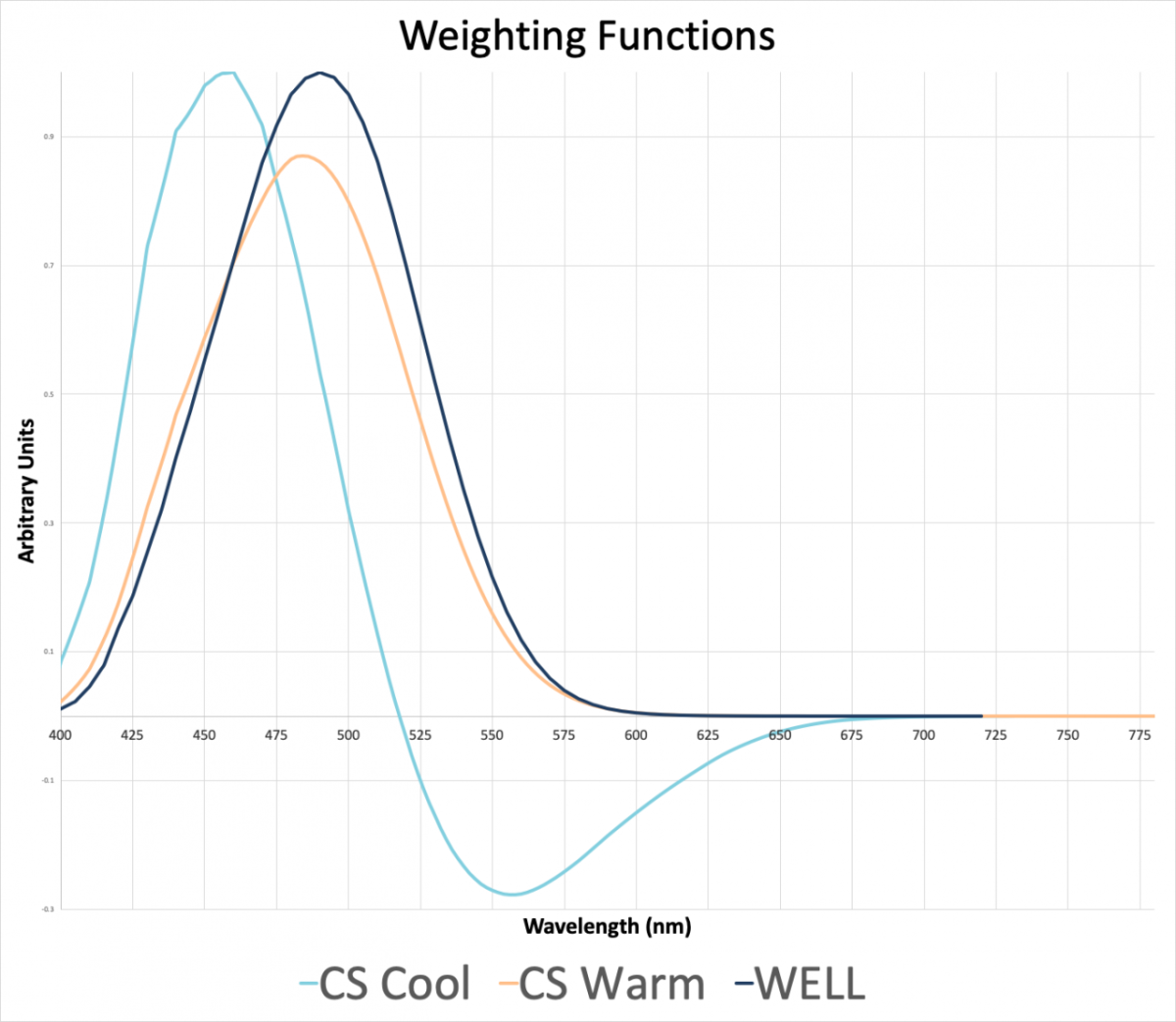
The melanopic lux model, as referenced in Feature L03: Circadian Lighting Design in WELL v2, has a peak sensitivity at 490 nm, which corresponds to light in the blue-green part of the spectrum (see Figure 1). (For comparison, the visual lighting metrics Wof lux or foot-candels are weighted at a peak response of 555 nm, in the yellow-green. This means that a light source with a high blue-green content will result in higher EML than a light source at the same visual intensity with relatively more orange-red, as a result of the greater activation of the ipRGCs.)
The circadian stimulus model has two separate weighting functions, depending on the characteristics of the light. Warm light (<3500K) would use the “warm” weighting function and cool light (>3500K) would use the “cool” weighting model. The model will determine which weighting factor shall be applied. Each of these weighting curves appear on Figure 1 below. Note, the “warm” weighting function has a peak sensitivity of 485 nm, which is similar to the 490nm from the Melanopic lux model.
Calculations and dataTaking a database I created with 39 different LED spectra from major manufacturers (see supplemental data here for details and an example calculation for the two models), I applied each light source to the melanopic lux model and the circadian stimulus model. From this data, we can calculate the vertical photopic lux and vertical foot-candles required to meet the daytime criteria for Feature L03: Circadian Lighting Design of the WELL Building Standard and the Lighting Research Center’s recommendations. These criteria/recommendations are as follows:
- WELL requires 150 melanopic lux for a minimum of 4 hours per day from electric light alone.
- The Lighting Research Center recommends a CS≥ 0.3, which corresponds to the ability to suppress melatonin by 30% [18].
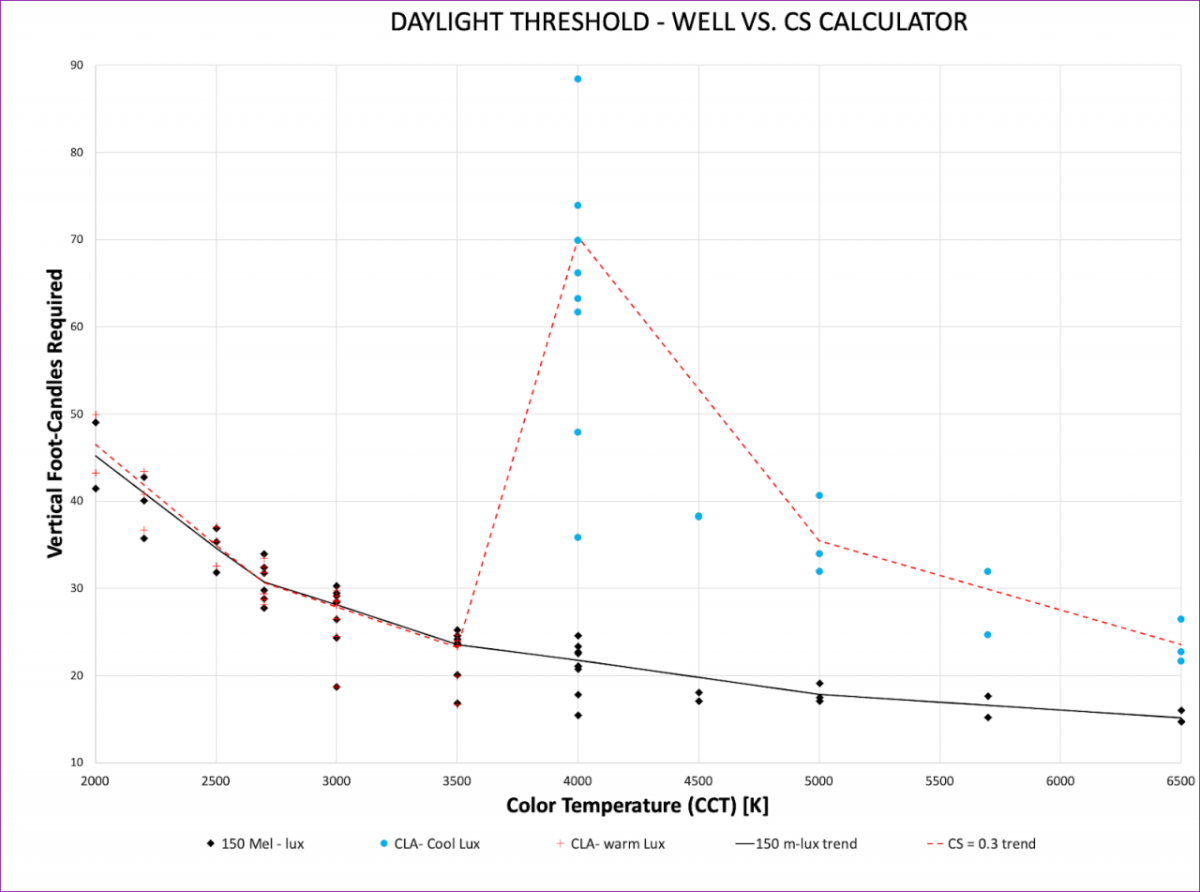
Figure 2 illustrates the foot-candle requirements to achieve 150 melanopic lux for all of the spectra from the 39 different LED sources of varying color temperature and color rendering, where each data point is a singular answer. Data was categorized by each product’s listed color temperature (i.e. 3000K, 3500K, 4000K, etc.). Trend lines are an average of each of those individual data points.
From the data in Figure 2, we notice a few things:
- For 3500K and lower (warmer), the amount of vertical foot-candles required to achieve a CS = 0.3 is practically identical to the foot-candles required to achieve 150 Melanopic lux.
- At 3500K, we need approximately 24 vertical foot-candles to meet both 150 Melanopic lux and a CS = 0.3. This represents the lowest light intensity where both criteria from WELL and LRC are satisfied.
- The CS transition from the “warm” to the “cool” weighting function creates a large discontinuity in the visual light levels required to achieve CS = 0.3.
- When applying the CS “cool” weighting function, it requires more vertical foot-candles to achieve a CS = 0.3 than it does to achieve 150 melanopic lux.
As we think about incorporating either model into lighting designs, at 3500K and warmer, it requires roughly the same number of vertical foot-candles to achieve a CS = 0.3 as it does to achieve 150 melanopic lux. Additionally, as we move toward cooler color temperatures, it requires significantly more vertical foot-candles to achieve a CS = 0.3 than it does to achieve the WELL criteria of 150 melanopic lux. Similar results are shown in the findings by IWBI’s Chief Engineer, Nathan Stodola, presented at the IES Light and Health conference in 2018 [19]. Design practices in this industry gravitate toward 3500K or warmer anyway, making this a convenient place to start. However, this target of 24 vertical foot-candles is higher than common designs in the United States, though spectral optimization of the light source allows for this target to be below 17 vertical foot-candles, showing that designing for circadian impact and energy conservation do not have to be at odds with each other.
The principle of “first, do no harm” has been adopted by many lighting designers regarding this topic as rationale to ignore circadian impacts. However, evidence of the myriad of deleterious health risks associated with circadian disruption, and the fact that 87% of people have circadian disruption, suggest that we are, in fact, doing harm with today’s lighting practices. I am motioning that we, as an industry, reconsider this phrase by saying “first, do less harm”.
Finally, it should be noted that there are some who believe both models are not sufficiently complete for determining circadian effectiveness. However, the biological pathways are identified, the circuitry fairly well understood [20], and the problem is real. While it is reasonable to believe that a more accurate model may present itself later down the road in the years to come, the first step to creating a solution is here now. Lets begin.
[1] Boyd, J. (2012). Plants use circadian rhythms to prepare for battle with insects. News.rice.edu. http://news.rice.edu/2012/02/15/plants-use-circadian-rhythms-to-prepare-for-battle-with-insects/
[2] Chapurlat, E., Anderson, J., Ågren, J., Friberg, M., & Sletvold, N. (2018). Diel pattern of floral scent emission matches the relative importance of diurnal and nocturnal pollinators in populations of Gymnadenia conopsea, 711–721. http://doi.org/10.1093/aob/mcx203
[3] Merrow, M., Spoelstra, K., & Roenneberg, T. (2005). The circadian cycle: daily rhythms from behaviour to genes. EMBO Reports, 6(10), 930–5. http://doi.org/10.1038/sj.embor.7400541
[4] Zarrinpar, A., Chaix, A., & Panda, S. (2015). Daily Eating Patterns and Their Impact on Health and Disease. Trends in Endocrinology & Metabolism, xx(2), 1–15. http://doi.org/10.1016/j.tem.2015.11.007
[5] Brown, T. M. (2016). Using light to tell the time of day: sensory coding in the mammalian circadian visual network. The Journal of Experimental Biology, 219(12), 1779–1792. http://doi.org/10.1242/jeb.132167
[6] Berson, D. M. (2002). Phototransduction by Retinal Ganglion Cells That Set the Circadian Clock. Science, 295(5557), 1070–1073. http://doi.org/10.1126/science.1067262
[7] Gaggioni, G., Maquet, P., Schmidt, C., Dijk, D., & Vandewalle, G. (2014). Neuroimaging, cognition, light and circadian rhythms. Frontiers in Systems Neuroscience, 8(July), 126. http://doi.org/10.3389/fnsys.2014.00126
[8] Espiritu, R. C., Kripke, D. F., Ancoli-Israel, S., Mowen, M. A., Mason, W. J., Fell, R. L., … Kaplan, O. J. (1994). Low illumination experienced by San Diego adults: Association with atypical depressive symptoms. Biological Psychiatry, 35(6), 403–407. http://doi.org/10.1016/0006-3223(94)90007-8
[9] Zeitzer, J. M., Dijk, D., Kronauer, R. E., Brown, E. N., & Czeisler, C. A. (2000). Sensitivity of the human circadian pacemaker to nocturnal light : melatonin phase resetting and suppression, 695–702.
[10] Roenneberg, T., & Merrow, M. (2016). The Circadian Clock and Human Health. Current Biology, 26(10), R432–R443. http://doi.org/10.1016/j.cub.2016.04.011
[11] Roenneberg, T., Allebrandt, K. V., Merrow, M., & Vetter, C. (2012). Social jetlag and obesity. Current Biology, 22(10), 939–943. http://doi.org/10.1016/j.cub.2012.03.038
[12] Sulli, G., Manoogian, E., Taub, P., Panda, S. (2018). Training the Circadian Clock, Clocking the Drugs, and Drugging the Clock to Prevent, Manage, and Treat Chronic Diseases. Trends Pharmacol Sci. 2018 Sep;39(9):812-827. doi: 10.1016/j.tips.2018.07.003.Epub 2018 Jul 27.
[13] Skeldon, A. C., Phillips, A. J. K., & Dijk, D.-J. (2017). The effects of self-selected light-dark cycles and social constraints on human sleep and circadian timing: a modeling approach. Scientific Reports, 7(September 2016), 45158. http://doi.org/10.1038/srep45158
[14] Lucas, R. J., Peirson, S. N., Berson, D. M., Brown, T. M., Cooper, H. M., Czeisler, C. A., … Brainard, G. C. (2014). Measuring and using light in the melanopsin age. Trends in Neurosciences, 37(1), 1–9. http://doi.org/10.1016/j.tins.2013.10.004
[15] Brainard, G. C., Hanifin, J. P., Greeson, J. M., Byrne, B., Glickman, G., Gerner, E., & Rollag, M. D. (2001). Action spectrum for melatonin regulation in humans: evidence for a novel circadian photoreceptor. The Journal of Neuroscience : The Official Journal of the Society for Neuroscience, 21(16), 6405–12. http://doi.org/21/16/6405 [pii]
[16] Figueiro, M. G., Bierman, A., & Rea, M. S. (2008). Retinal mechanisms determine the subadditive response to polychromatic light by the human circadian system. Neuroscience Letters, 438(2), 242–245. http://doi.org/10.1016/j.neulet.2008.04.055
[17] Schlangen, Luc. (2018). CIE international Standard (CIE S 026/E:2018) “System for Metrology of Optical Radiation for ipRGC-Influenced Responses to Light” http://www.cie.co.at/publications/international-standards.
[18] Figueiro, M. G., Steverson, B. C., Heerwagen, J., & Rea, M. S. (2014). Circadian light and its impact on alertness in office workers : A field study, 1–13.
[19] Stodola, N. (2018). Mathematical Comparison of Circadian Lighting Metrics. Paper presented at Light + Human Health: Circadian, biological + behavioral IES Research Symposium, Atlanta, Georgia. https://www.ies.org/product/light-human-health-circadian-biological-behavioral-2018-ies-research-symposium/
[20] Hughes, S., Jagannath, a, Rodgers, J., Hankins, M. W., Peirson, S. N., & Foster, R. G. (2016). Signalling by melanopsin (OPN4) expressing photosensitive retinal ganglion cells. Eye, 44(November 2015), 1–8. http://doi.org/10.1038/eye.2015.264